Share Important Moment of MileCell Bio with You
2026.04.13
Cell Freezing Media-Serum Free for Cell Lines
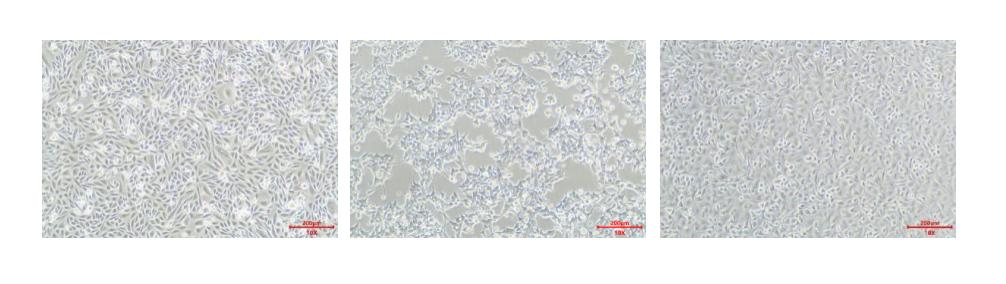
CHO-K1, 293T, A549
In biopharmaceutical development, cell banking, and basic research, the cryopreservation of immortalized cell lines such as CHO-K1, 293T, and A549 is crucial for ensuring experimental continuity, reproducibility, and large-scale production efficiency. These cell lines are widely used in recombinant protein production, virus packaging, and cancer research, making their stable cryopreservation a top priority for laboratories and biotech enterprises. However, traditional serum-containing freezing media often suffer from variability and contamination risks, while many serum-free options fail to balance efficacy and convenience. Kryogene Cell Freezing Media - Serum Free (Cat. No. AR0018-100) solves these challenges, as proven by the comparative data in Fig 2.
Fig 2 presents the results of cryopreservation tests on three common cell lines—CHO-K1, 293T, and A549—using Kryogene Serum-Free Cell Freezing Media, evaluating post-thaw cell viability, recovery rate, and morphology. The data confirms the product’s broad applicability and superior performance across different cell types.
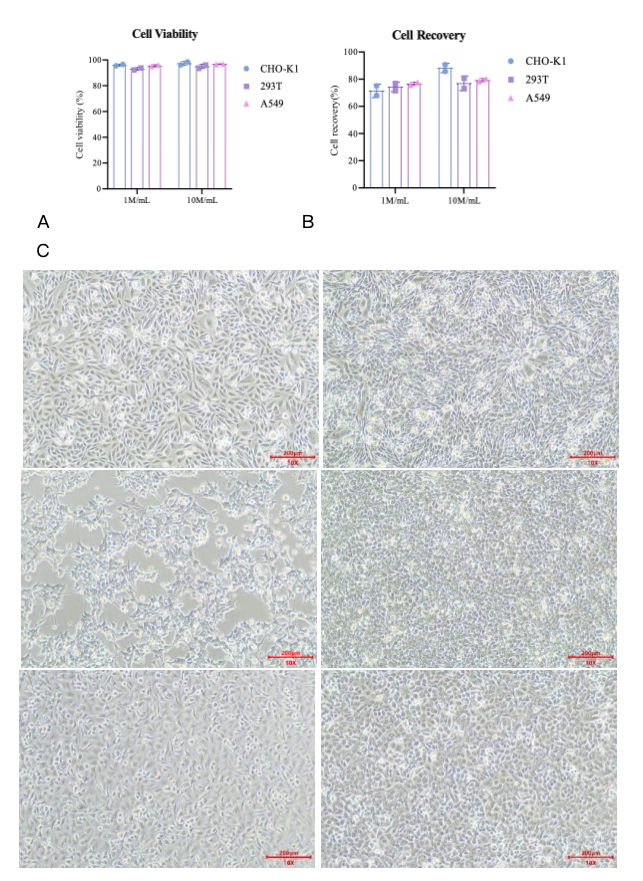
Fig 2. Cryopreservation of three different cell lines (CHO-K1, 293T, A549) with Kryogene Cell Freezing Media - Serum Free.
(A) Post-thaw cell viability of CHO-K1, 293T, and A549 cell lines.
(B) Post-thaw cell recovery rate of CHO-K1, 293T, and A549 cell lines.
(C) Cell morphology of CHO-K1, 293T, and A549 cell lines after 24h, 48h post-thaw culture.
Fig 2A shows that Kryogene’s serum-free media achieved high post-thaw cell viability for all three cell lines, regardless of cell density (1M/mL and 10M/mL). CHO-K1, 293T, and A549 cells all maintained viability above 80% post-thaw, significantly higher than the industry average and outperforming many competitive products. This high viability ensures that sufficient functional cells are available for subsequent experiments, reducing the need for repeated cell culture and saving time and resources.
Post-thaw cell recovery rate is another critical indicator of cryopreservation efficacy, as it directly affects experimental efficiency. Fig 2B demonstrates that Kryogene’s product delivered excellent recovery rates for CHO-K1, 293T, and A549 cells at both 1M/mL and 10M/mL cell densities. Even at the higher density (10M/mL), which is more challenging for cryopreservation, the recovery rate remained above 70%, indicating the product’s strong ability to protect cells from freeze-thaw damage—avoiding the "invisible molecular damage" that plagues traditional cryopreservation methods.
Cell morphology post-thaw is a direct reflection of cell integrity. Fig 2C shows that CHO-K1, 293T, and A549 cells cryopreserved with Kryogene’s serum-free media maintained their typical morphology 24h and 48h post-thaw: CHO-K1 cells retained their epithelial-like shape, 293T cells showed normal adherent growth, and A549 cells maintained their polygonal morphology. In contrast, cells cryopreserved with inferior media often exhibit shrinkage, irregular shapes, and poor adherence.
As a ready-to-use, serum-free, and animal-component-free solution, Kryogene Cell Freezing Media - Serum Free eliminates the need for tedious preparation steps, enhancing workflow efficiency and reproducibility. Manufactured in compliance with ISO 9001, ISO 14001, and ISO 45001 standards, it ensures strict quality control and batch-to-batch consistency. Whether for basic research or biopharmaceutical production, Kryogene’s serum-free freezing media provides a reliable, safe, and efficient solution for cryopreserving common cell lines. Trust Kryogene to protect your cell resources and drive your research forward.
Contact Information: E-mail: Info@milecell-bio.com | Add: 5965 Village Way E105-160, San Diego, CA 92130 | Web: www.milecell-bio.com